©Copyright 2018 GEOSCIENCE RESEARCH INSTITUTE
11060 Campus Street • Loma Linda, California 92350 • 909-558-4548

THE CHEMICAL COMPOSITION OF THE EARTH'S ORIGINAL ATMOSPHERE
by
John C. Walton
Lecturer in Chemistry
University of St. Andrews, Fife, Scotland
Related page | REACTION |
It has often been postulated that the original atmosphere of the earth was a reducing one. Such an atmosphere, unlike the present oxidizing kind, would facilitate the formation and survival of those molecules believed to be associated with the spontaneous origin of life. The author discusses some of the cosmological, geological, and biological evidence related to this important issue.
According to a popular theory of the origin of life, which has attained almost the status of orthodoxy in the world of science, a variety of physico-chemical processes led to the formation and accumulation of biologically important, simple organic compounds early in the earth's history. These molecules combined together to give more and more complex products until one was formed that could be called living [1]. Oxygen rapidly degrades the majority of organic compounds and inhibits many of the metabolic reactions in living systems, and therefore the original proponents of this theory postulated that the primitive earth must have had a reducing atmosphere devoid of oxygen [2], [3].
In 1953 Miller carried out his famous experiments in which reducing atmospheres of methane, ammonia, water and hydrogen were subjected to electric discharges and a wide variety of organic compounds, including amino acids, were formed [4]. Numerous experiments purporting to simulate primitive earth conditions with a variety of gas mixtures and energy sources have subsequently confirmed that amino acids and other bio-organic molecules are synthesized under reducing conditions [5], [6], but not in the presence of oxygen [6]- [9]. Bio-organic compounds are not forming in significant quantities in the earth's present atmosphere or hydrosphere (except in living organisms) and therefore the initial steps in this mechanism for the origin of life depend crucially on the nature and composition of the earth's original atmosphere. In this article the cosmological, thermodynamic, and geological evidence bearing on this question is reviewed. It is found that the facts are in better agreement with an early oxidising atmosphere similar to the present one.
A. FEASIBILITY AND STABILITY OF A REDUCING ATMOSPHERE
1. Cosmological Evidence
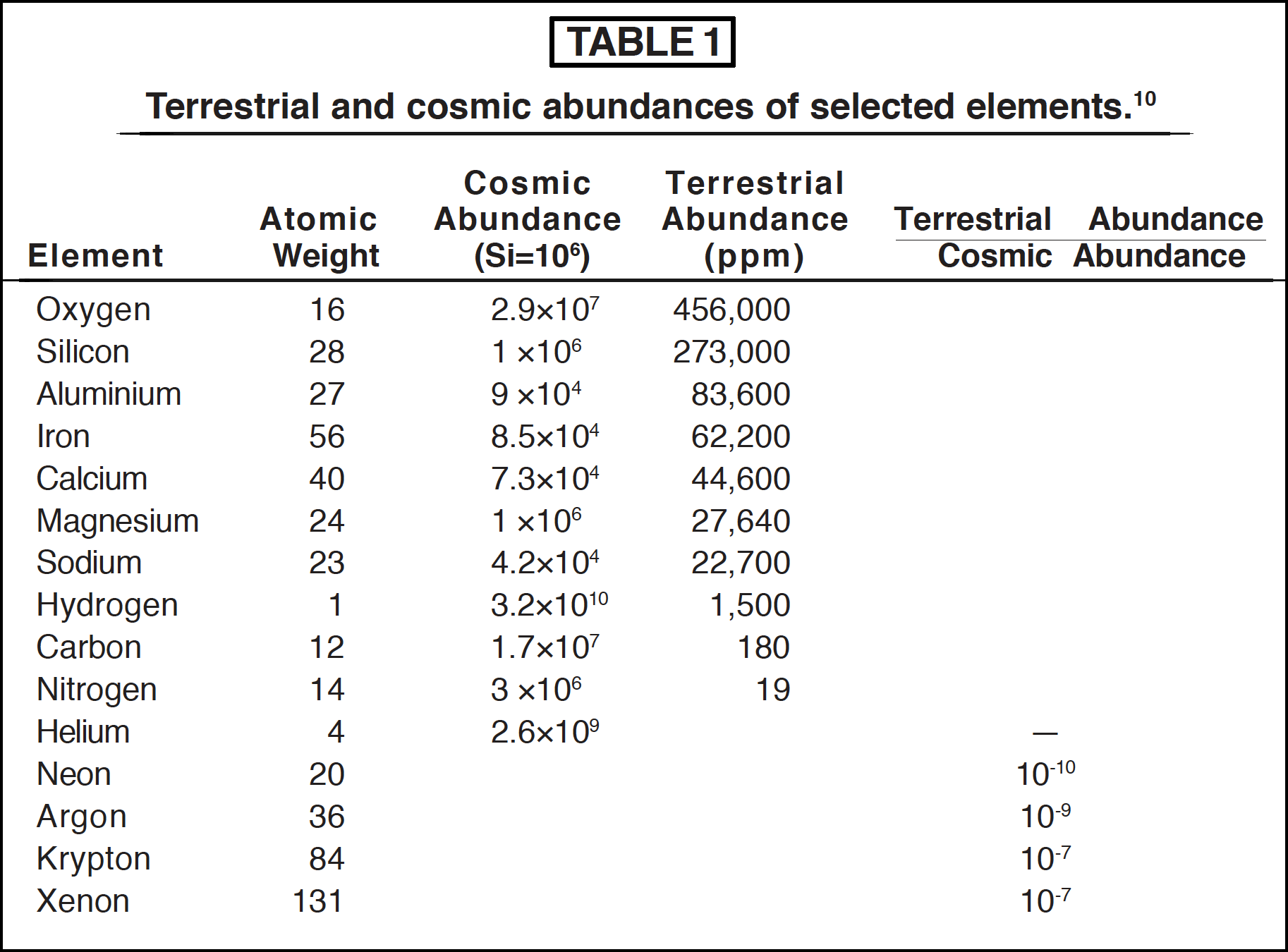
In the most generally held theory of the origin of the solar system, the sun and planets are supposed to have condensed from a dust cloud having the same average composition as the universe. The cosmic abundances of a selection of elements are given in Table 1, which shows that hydrogen and helium are by far the most abundant.
Sources: see ref. [10]
Early workers in this field therefore concluded that the earth must originally have had an atmosphere rich in hydrogen and the reduced compounds of other elements such as ammonia, methane and water, somewhat similar to the atmosphere known to exist on the heavy planets such as Jupiter and Saturn, but containing less helium and hydrogen as these light elements would escape from the earth's gravitational field. A major difficulty with this view is the relatively low amount of the rare gases on the earth compared to their cosmic abundances. The ratios of the cosmic abundances of the rare gases to their terrestrial abundances are given in Table 1, and it is apparent that the earth contains many orders of magnitude less neon, argon 36, krypton and xenon than it should if it were condensed from average cosmic matter.
This view has now largely been abandoned in favour of the idea that volatile elements such as hydrogen were lost from the earth during the condensation stage while the gravitational field was weak. The earth's reducing atmosphere is then supposed to have accumulated by outgassing of the interior by volcanic and allied activity. It is difficult to accept that xenon and krypton with atomic weights of 131 and 84 respectively could be so thoroughly lost from this early gravitational field, and yet methane, ammonia and water, and a host of lighter compounds be retained. Even if the additional hypothesis that these molecules were retained in chemically combined form, such as carbonates and ammonium salts, is advanced, it would still be expected that much of the rare gases should be retained by occlusion within dust particles and larger fragments composing the cloud. Argon, for example, is trapped with fair efficiency in rocks, and this is an important factor in the potassium-argon dating technique. The extremely low terrestrial abundances of the rare gases are still, to say the least, surprising. The chemically combined forms of carbon and nitrogen so far detected in cosmic dust clouds are volatile, low molecular weight compounds such as formaldehyde, hydrogen cyanide, methanol and acetaldehyde [11]. The most natural conclusion from this is that the earth's composition has always been unlike the average of the universe.
Gases evolving from present-day volcanoes vary in composition with locality and time. Typical compositions of the emanations from some volcanoes and fumaroles are shown in Table 2.
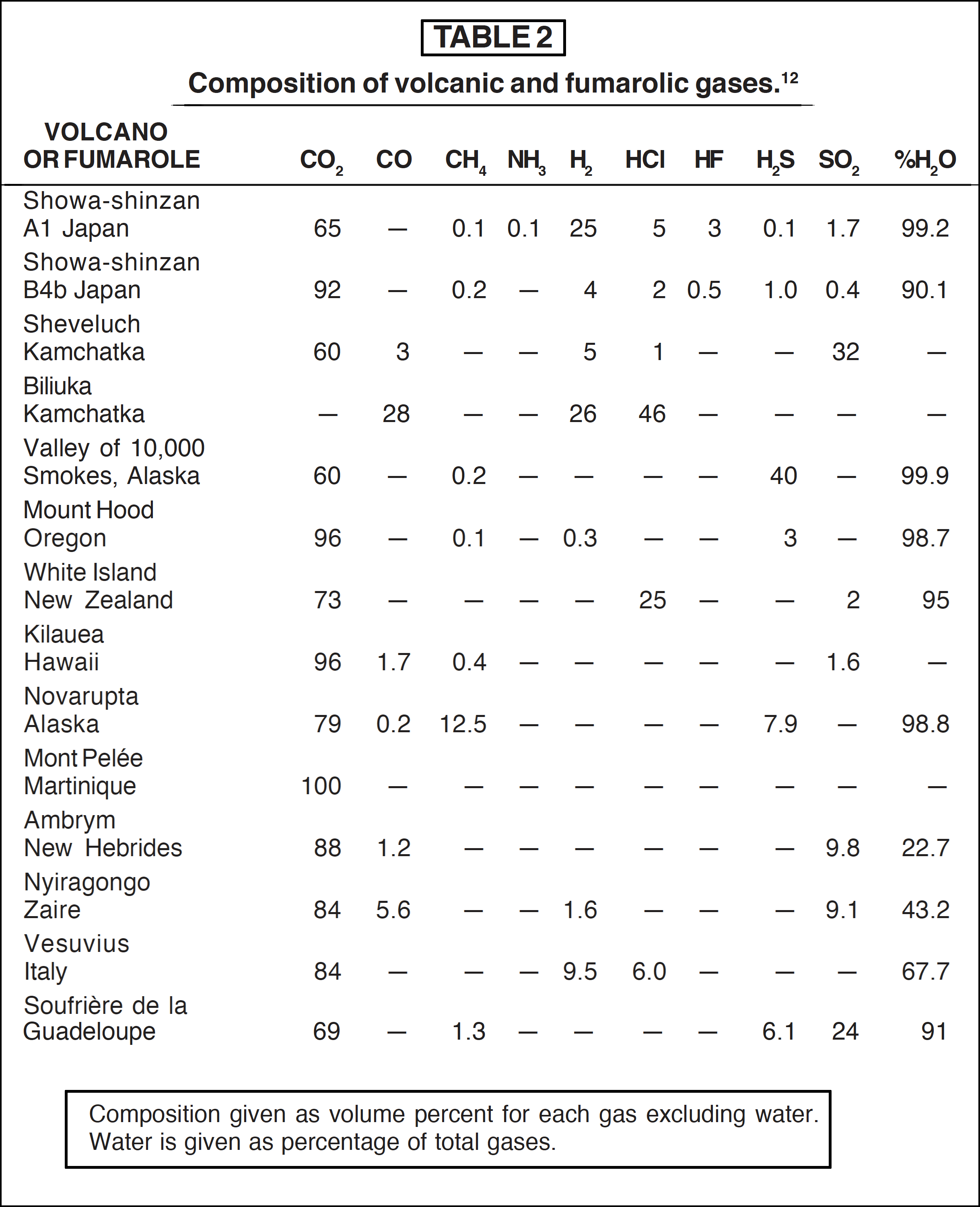
Steam and carbon dioxide are usually the main constituents, but occasionally hydrogen, carbon monoxide, sulphur dioxide or hydrogen chloride may be important. Methane and ammonia are rarely found, and then usually in trace quantities only. The hypothesis that the earth acquired a reducing atmosphere by volcanic outgassing finds little support in the evidence from present-day volcanoes. The hypothesis can be bolstered by making the additional assumption that volcanic gases were much more reducing in the past [13]. This sheds no further light on the problem since it is merely the complement of the original reducing atmosphere hypothesis. It is interesting to note that Precambrian igneous rocks closely resemble their modern counterparts in chemical composition (see below).
2. Kinetic and Thermodynamic Considerations
Can molecules such as methane, ammonia, or carbon monoxide in contact with the ocean, the earth's crust, and in the presence of sunlight persist for geologically significant periods of time? Methane is the thermodynamically stable carbon compound in the presence of excess hydrogen:
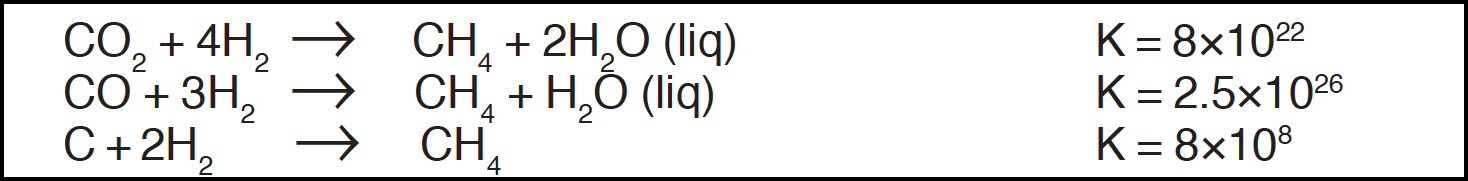
In the absence of hydrogen, however, ultraviolet light and electric discharges readily decompose methane, which also reacts with water to give carbon dioxide and carbon monoxide. Carbon monoxide reacts quite rapidly with ocean water (pH 8.1) to give formate:

and it is unlikely that carbon monoxide could persist even in a reducing atmosphere.
Ammonia is formed from nitrogen and hydrogen in a fairly favourable process:

but it is also rapidly destroyed by ultraviolet radiation; so that a quantity of ammonia equivalent to the present atmospheric nitrogen would be degraded in this way in 30,000 years [14]. Ammonia is also highly soluble in water and would dissolve in the ocean to form ammonium ions thus effectively being removed from the atmosphere [15].
The indications are that carbon monoxide and ammonia could not persist in the atmosphere in contact with the ocean, and the availability of methane depends on a constantly supplied reservoir of hydrogen in the atmosphere. At present there is relatively little hydrogen available in the earth's crust (see Table 1). According to the dust cloud model for the formation of the earth, the majority of the hydrogen and other volatiles would have been lost before consolidation of the earth [16]. Any hydrogen surviving this process, or being produced by photodissociation of water vapour or by volcanic emanation, is also rapidly lost from the earth's gravitational field because of its low molecular weight.
The exact rate of loss of hydrogen is difficult to assess, since it depends on the rate of diffusion of hydrogen to the escape layer, and the temperature and diameter of the exosphere, none of which can be known for the primitive earth. At present the temperature of the escape layer is extremely high (~1500ºK) so that almost all hydrogen getting there escapes. It seems doubtful that hydrogen (and hence methane, ammonia or other reduced gases) could ever have reached substantial concentrations in the atmosphere, and in view of this the formation and persistence of a reducing atmosphere must be considered unlikely under any geologically plausible conditions.
One of the most significant facts in this area, which is often overlooked, is the great abundance of oxygen available on the earth. It is by far the most abundant element in the earth's crust (see Table 1) which is largely composed of oxides of silicon, calcium, aluminium and other metals [17]. Since oxygen is also very abundant in the universe, being third in magnitude after hydrogen and helium, it is probable that very great amounts must have been present from the beginning, regardless of the mechanism for the formation of the earth.
Furthermore, oxygen is also overwhelmingly abundant combined with hydrogen in the hydrosphere.
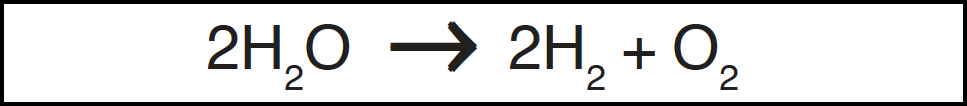
If we assume for the moment that oxygen had been absent from the atmosphere at some time, then several mechanisms exist which would rapidly build up its concentration. Water vapour is dissociated by ultraviolet radiation in the upper atmosphere to give hydrogen, most of which escapes into space, and oxygen which is retained by the earth's gravitational field:
Urey [18] has suggested that production of oxygen in this way would be limited because the oxygen formed would absorb the ultraviolet light needed to photodissociate the water. This self-regulating mechanism depends on the difference in vertical distribution of oxygen and water in the atmosphere. Oxygen is distributed exponentially, but water is arrested in a cold trap in the troposphere. Oxygen would rise much higher in the atmosphere, and since oxygen and water absorb radiation substantially in the same region of the spectrum, the water would be screened from further dissociation as soon as sufficient oxygen built up [19]. Berkner and Marshall [20] presented detailed calculations which showed that this mechanism would limit oxygen in the atmosphere to about one thousandth of its present atmospheric level. Brinkmann, however, reconsidered this problem and showed that Berkner and Marshall's calculations were invalid because they ignored the pathlength dependence of the oxygen absorption and the dissociation of water when it was a minor absorber [21]. Brinkmann showed that the Urey self-regulation mechanism is much less effective than was formerly supposed, and that oxygen could have reached an appreciable fraction of the present atmospheric level from water dissociation alone. He concludes that the atmosphere could have been oxidising over a large fraction of geologic time. Direct observations from the moon during the Apollo 16 mission revealed that substantial amounts of hydrogen are leaving the earth's atmosphere, due to photochemical water dissociation in the upper atmosphere [73].
A second important mechanism for the production of oxygen is photosynthesis by living organisms. The rate of oxygen production is extremely high, and it is estimated, for example [22], that at the present time all the oxygen in the atmosphere passes through the photosynthetic cycle in 2000 years, and all the carbon dioxide in 300 years. The oldest known rocks contain evidence of photosynthetic organisms, although this is by no means conclusive (see below). Van Valen has pointed out that when oxygen is produced in photosynthesis, a stoichiometrically equivalent quantity of reduced carbon is also formed [22]. If oxygen had built up from a very low concentration to the present atmospheric level, there should be an accumulation of reduced carbon in the earth equivalent to far more than the reserves of coal and oil now known. Van Valen considers that there is no satisfactory mechanism for regulating oxygen concentrations at very low levels.
When sedimentary rocks are subjected to high pressures and temperatures in metamorphic processes in the earth's crust, the dominant chemical process occurring is progressive loss of water:
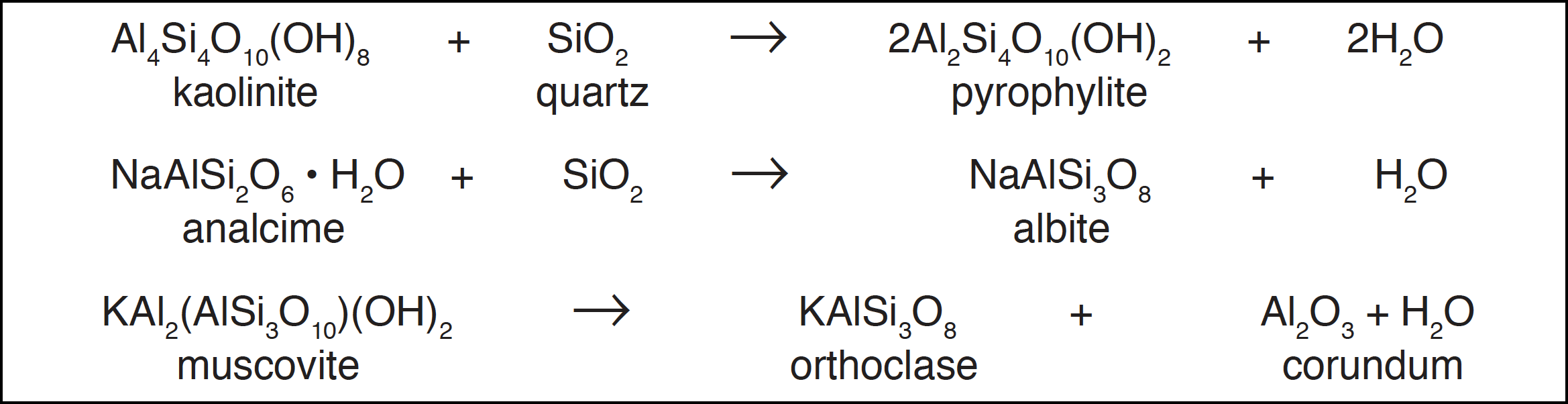
With carbonate minerals carbon dioxide can be formed, and oxygen can also be produced in gas-solid reactions involving abundant minerals:
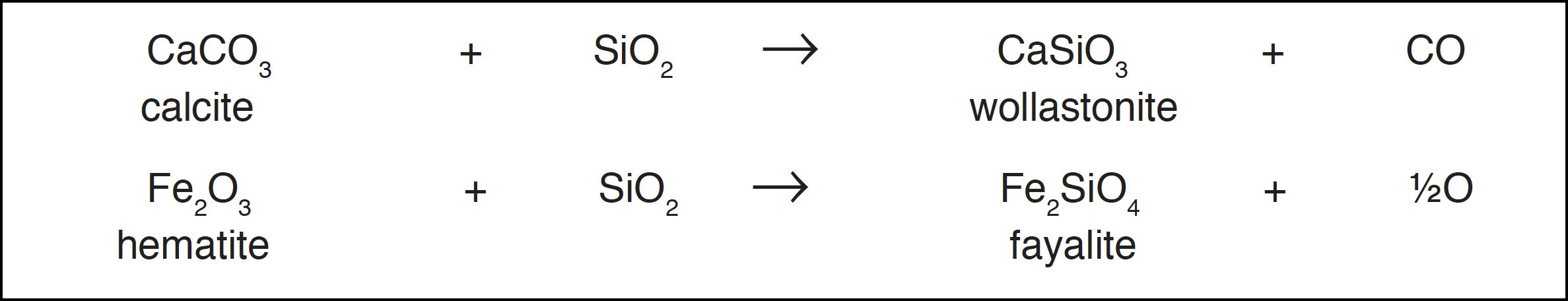
This process is probably not important as a direct contributor to atmospheric oxygen, since volcanic gases rarely contain oxygen, but the dominant position of water and carbon dioxide in volcanic emanations is readily explained. A facile route is seen to exist for the transfer of oxygen from the vast reservoirs in the rocks to the ocean where it can be used in photolysis or photosynthesis and thence to the atmosphere.
B. GEOLOGICAL EVIDENCE
It might be expected that conclusive evidence regarding the earth's original atmosphere could be obtained from a study of the oldest known rocks in the Precambrian formations. Unfortunately, Precambrian rocks have suffered extensively from metamorphic and diagenetic processes which have altered their original form and content chemically and physically. Most of the exposed rocks have suffered so much that most of the evidence of their early history has been destroyed. An additional problem is the contamination of the ancient rocks with material of recent origin carried in by circulating ground water or processes of diffusion. Interpretation of the evidence is a highly subjective process and, not surprisingly, geologists are divided amongst several schools of thought. Rutten has presented the position favouring an early reducing atmosphere very persuasively in his book The Origin of Life by Natural Causes [23]. Another school of thought favours the view that the atmosphere has been oxidising since the formation of the earliest crustal rocks [24].
1. Oxidation State of Iron in Precambrian Rocks
Evidence commonly cited as favouring an early reducing atmosphere is the preponderance of the reduced form of iron in Precambrian formations. Rankama, for example, reported that ferrous oxide predominated over ferric oxide in Precambrian conglomerates from Suodenniemi, Finland [25]. Nanz also found a preponderance of ferrous iron in Precambrian slates from Michigan and Wisconsin [26]. The fact that oxidised forms of iron occur at all in the Precambrian might be taken to indicate an oxidising atmosphere. Even the oldest known sedimentary rocks from the Isua iron formation in Greenland contain bands of magnetite (Fe3O4) of intermediate oxidation state [27]. Vast quantities of magnetite and hematite are present in Precambrian iron formations, which would require an immense amount of oxygen for their formation from reduced iron compounds. Where could all this oxygen come from if not directly or indirectly from the atmosphere [28]?
The preponderance of ferrous iron might seem significant until it is recognized that metamorphic processes often lead to partial reduction of iron [26] and that many reducing environments are known on the earth at the present day. Reducing conditions prevail deep in the earth's crust, and materials brought to the surface by volcanic activity or rising hydrothermal waters are commonly in reduced form. Fyfe suggested that the ferrous oxide in Precambrian rocks is simply a reflection of the enhanced volcanism prevailing in earlier times [24]. Reducing conditions also hold sway in deep waters such as the Norwegian fjords and the Black Sea [29] and in the northern Indian Ocean [30]. Throughout geologic time and even today, ferrous iron is effectively transported and deposited by subsurface waters [31]. The fact that all oxidation states of iron, from hematite to magnetite to siderite (FeCO3) to pyrite (FeS2), have been found in sediments of all ages merely indicates that the oxidation state of sediments depends primarily on local conditions that do not reflect the average atmospheric composition at the time of deposition.
2. Uraninite and Pyrite in Precambrian Gold-Uranium Reefs
Precambrian deposits in the Dominion and Witwatersrand Reefs, South Africa; Serra de Jacobina, Brasil; and Blind River, Ontario, districts contain ancient conglomerates and sands cemented to a very hard rock carrying grains of uraninite (UO2), pyrite and ilmenite (FeTiO3). The origin of these gold-uranium blanket ores has been controversial for more than half a century. In 1958 Ramdohr suggested they could be considered as sediments laid down under an anoxygenic atmosphere and presented evidence for repeated cycles of weathering-erosion-transportation-sedimentation, indicating that these beds must have been in repeated contact with the contemporaneous atmosphere. The reduced state of the pyrite and uraninite (the stable form of uranium under present-day atmospheric conditions is UO3) was taken as an indication of an anoxygenic atmosphere at the time of deposition [32], [23]. Davidson, on the other hand, maintains that the uranium and pyrite ore bodies have been formed within the earth's crust long after the mother rock sediments were laid down, by infiltration of hydrothermal metal-bearing solutions, and that they cannot be explained in terms of abnormal atmospheric conditions [24], [28]. Even Rutten, an enthusiastic supporter of the reducing atmosphere hypothesis, admits that the deposits show extensive effects of hydrothermal processes [33].
If, however, it could finally be proved that the uraninite and pyrite deposits are sedimentary in origin, there is no need to invoke an anoxygenic atmosphere to explain their formation. The thermodynamically stable uranium oxide under an atmosphere of oxygen is UO3, and consequently UO2 converts to this, but the rate of conversion depends strongly on the physical form of the uraninite. Conversion is rapid for finely divided or powdered UO2, but granular or compacted material is stable indefinitely [34] and it is even used, fabricated into rods, as fuel elements for nuclear reactors. Grains of uraninite would be expected to remain unchanged during erosion, transportation and deposition even under the present oxygenic atmosphere. Zeschke has shown that uraninite is transported as minute grains by the Indus River in Pakistan at the present day 35. Furthermore, in the Mozaan rocks of Swaziland, deposited concurrently with the Witwatersrand strata, and in the Lorrain sandstones of Ontario, almost contemporary with the Blind River conglomerates, heavy mineral assemblages, completely of modern aspect, are widely developed [24]. This indicates that the uranium-pyrite ores, which are themselves strikingly similar to recent deposits except for the presence of uraninite and pyrite grains [36], cannot be explained in terms of abnormal atmospheric conditions.
Many of the Precambrian mineral deposits are associated with carbonaceous matter. Uraninite and pyrite are frequently found in association with thucolite, a carbonaceous mineral, and kerogen, which is graphitized organic matter resembling medium- to high-grade coal [37], [38]. Pyrite is deposited in present-day environments, such as the Black Sea, by the action of bacterial sulphate-reducers [39]. Micro-organisms are known to play an important role in the deposition and concentration of many minerals. The presence of carbonaceous matter in Precambrian ore deposits has generated interest in the possibility that they owe their formation to oxygen-producing or oxygen-utilising micro-organisms [40]. According to Koen the uranium was fixed and concentrated by biological action [41], and it has been observed that the gold-uranium reefs of the carbon seam type of the Kaapvall Craton are confined to environments expected to favour algal growth [42]. Schidlowski, on the other hand, has advocated an origin for the thucolite involving migration of biogenic compounds into the conglomerates where they are exposed to radiation from the uranium and undergo condensation and polymerization reactions, finally solidifying as carbonaceous material around individual uraninite grains (37). Doubt has been cast on this hypothesis by observations on Precambrian thucolite occurrences in Australia which showed that the substance does not have the properties expected of a radiolytic polymer [43].
Obviously no final conclusions can be drawn at present, but there must be a strong presumption that photosynthetic organisms are involved, particularly for pyrite. Further support for this view comes from the isolation of chemical fossils including porphyrins from the carbonaceous material (see below).
3. Precambrian Banded Iron Formations
Another type of rock which is supposed to provide evidence of an anoxygenic atmosphere is the banded iron formation; composed of thin laminae of finely crystalline silica alternating with thin laminae of iron ore, the iron being mainly in the ferrous state. Cloud has repeatedly championed the view that the banded iron formations, which are most common in the middle Precambrian, represent unoxidised sediments laid down under a reducing atmosphere and that as the atmosphere became more oxidising towards the end of the middle Precambrian, the banded iron formations cease and are replaced by oxidised sediments known as red beds [44], [23].
There are many difficulties associated with this view. Even the most ancient banded iron formations contain hematite and magnetite [27], which were at least partly laid down in the primary deposition of the formations [45]. Fully oxidised minerals also occur in formations contemporary with the banded iron formations. Bedded iron ores in the Pretoria series of the Transvaal system (middle Precambrian) are mainly hematite-chamosite or magnetite-chamosite oölites akin to the Wabana ores of Newfoundland and other Phanerozoic formations [28], which clearly contradicts the hypothesis of an anoxygenic atmosphere. There is no clear distinction in time between the cessation of banded iron formations and the onset of red bed formation. Banded iron formations of late Precambrian and even younger age are known, although they are not common [45],[31]. A minority of geologists also consider that the banded iron formations are of volcanic origin [46], their reduced state having no direct connection with atmospheric conditions.
The majority view is that the deposits are sedimentary in origin, but the orthodox explanation of the thin laminations of iron ore does not require the presence of an anoxygenic atmosphere. It is proposed that weathering processes bring the more soluble ferrous iron down to deep lakes where it is trapped in solution in the deeper oxygen-free waters. Ferrous iron is then precipitated as soon as mixing of the waters brings oxygen to the lower water body [47], [48]. Environments such as this are common in present-day lakes.
Red beds are sandstone, siltstone or mudstone made of detrital grains set in a reddish-brown matrix or cemented by precipitated ferric oxide. They occur in great variety and complexity, and their origin and mode of formation have been something of an enigma for years [49]. Red beds are in fact known from the middle and early Precambrian, contemporary with the banded iron formations, although they are not common. Red beds are found in the Uyansk and Uchursk series of the Siberian platform, in the Roraima formation, South America, in the Lorrain formation of the Huronian in Ontario and possibly also in the much older Muruwa formation of Guiana [28].
Most of the oxidised iron in red beds is not considered to be an original constituent of the rock but to have been produced in situ by post-positional chemical changes [47], [48]. There is some evidence that the relative lack of red beds in the early Precambrian is because corresponding deposits are in the process of metamorphism whereby the red pigment is dissolved out by ground waters containing magnesium, and the mineral chlorite is then precipitated [28].
4. Geological Evidence of an Early Oxygenic Atmosphere
The proponents of an early reducing atmosphere have focused attention on certain special features of Precambrian strata considered above, but this evidence needs to be evaluated in the light of the overall situation in the Precambrian. The oldest rocks on earth are rather similar to modern ones, the sedimentary rocks being "rather normal," indicating that the oceans at the time were rather similar to modern ones [50]. Much the same kinds of sedimentary rocks are found in Precambrian as in Paleozoic and younger rocks [51], [52]: they are water laid and imply the existence of a substantial hydrosphere and atmosphere for weathering processes [53]. Sedimentary rocks found in the Precambrian are lithologically comparable with those of younger age [54], [55], and igneous rocks of early Precambrian age closely resemble modern rocks in chemical composition [46]. Although the size of the ocean in earlier times remains a matter of debate, there is no evidence that sea water chemistry has ever changed substantially, and it is commonly assumed that its pH and composition have been constant from the earliest times [50], [56]. The atmosphere-hydrosphere-lithosphere interaction is an extremely complex one, and the similarities in ancient and modern rocks and sediments clearly imply an atmosphere little different from the present.
All minerals except oxides are unstable in chemical weathering under an oxygenic atmosphere. The ions derived from weathering when transported into areas of sedimentation recombine, mostly forming clay minerals. Clays are as abundant in the early Precambrian as at present [57]. In a reducing atmosphere, weathering is expected to be much slower and quite different in character, since feldspars, dark minerals, sulphides etc., would be chemically unaffected, and weathering would take place by minor physical processes such as frost splitting and sun blasting [58]. The maturity of many Precambrian sediments, the roundness of sand grains, and the well-worn pebbles and cobbles found in early Precambrian conglomerates demonstrate that weathering, erosion and sedimentation went on then as now. Pettijohn considers that such mature weathering could not have been accomplished in the absence of some kind of plant cover [72]. Modern lateritic soils require humic acid derived from vegetation, living in an oxygenic environment, for their development. Precambrian banded iron formations, which resemble lateritic soils, are thought by some to have been formed in a similar way. This view is supported by the finding of frequent intercalations of graphite, possibly of biological origin in the iron formations [46].
Limestone is produced at present by the growth of green algae in calcium-bicarbonate-rich ground waters in an oxygenic environment. Inorganic deposition of limestone in stalactite caves is also known, but this is normally almost negligible in sunlit areas. Limestone deposits, although not abundant, are found in early, middle and late Precambrian times. Some of these deposits may be biogenic; for example, those described from the Bulawayan system, of early Precambrian age, by MacGregor [59], [60] and the prolific development of similar structures in the middle Precambrian of Finland [61] which are entirely comparable with recent algal reefs.
Since limestone is known to be deposited by green algae living in an aerobic environment, one may propose that the Precambrian atmosphere, at the time of deposition of the limestone reefs, was also oxidising. Of course it is always possible to postulate that the metabolism of lime-depositing algae in the past must have been quite different [57]. The soundest procedure, however, is to utilise the modern analogy and work from the known present to the unknown past. Limestone deposition by anaerobic fermenting organisms, though possible, would be expected to produce material of quite different chemical and physical composition, whereas the Precambrian deposits are notably similar to modern equivalents.
Chert or flint is an extremely fine-grained silica which forms, at the present day, by the chemical action of sea water on the siliceous skeletons of plankton such as Diatoms and Radiolaria. It also forms by concretionary action in rocks after their emergence from the sea, and in lacustrine limestones. Enormous amounts of cherts occur in Precambrian strata such as the banded iron formations, where indirect evidence for a biological origin is provided by its association with organic matter, including in some cases actual microfossils [38].
Phosphorites are deposits which in modern examples of formation develop from phosphatic phytoplankton living by photosynthesis in the uppermost 50 m of the ocean. Many occurrences of Precambrian phosphorites are known, such as those from the Upper Krivoi Rog series of middle Precambrian age, or the deposits of Slyudganka in eastern Sayan and the even older phosphorites in the Baikal region, all from the U.S.S.R. [28]. The present-day mode of formation of cherts and phosphorites strongly suggests the Precambrian equivalents developed in an oxygenic atmosphere.
5. Macrofossils, Microfossils and Chemical Fossils
Considerable evidence has accumulated in recent years indicating that the carbonaceous matter found in the Precambrian strata is of biosynthetic origin. The nature of this material also suggests that some of it was derived from oxygen-utilising and oxygen-producing organisms. Great caution needs to be exercised in the evaluation of this data because of the problem of contamination. Recent studies with the finest grained cherts and shales from the Precambrian have shown that they are significantly permeable to the flow of fluids and that organic material of modern derivation can be carried into the rock matrix by circulating ground waters [62], [63]. At present there are no wholly sound criteria for distinguishing between organic material laid down with the host rock and contamination of recent origin.
A second problem concerns the genuineness of the microfossils, many of which have very simple forms such as spheres or rods. Inorganic artifacts such as colloidal salts after drying can give rise to similar structures, and artifacts composed of iron, silica and other materials could certainly occur in sediments. The microfossils are generally accepted as genuine, but the possibility of "pseudofossils" cannot be ruled out.
The only macrofossils that are of widespread occurrence in the Precambrian are stromatolites: metric-sized mounds with a characteristic laminated structure. These are reef-like remnants usually presumed to have been formed from precipitated mineral matter on the enlarging surfaces of microbial communities (usually dominated by blue-green algae) growing by photosynthesis [64]. Stromatolites are known from the Bulawayan group of early Precambrian age, and are widespread in the middle and late Precambrian. The fossil stromatolites bear a marked resemblance to modern ones forming at Hamelin Pool near Shark Bay, Australia [64], [65].
The Onverwacht and Fig Tree groups of the Swaziland system, South Africa, are considered to be the oldest relatively unmetamorphosed rocks known. Numerous globular, filamentous and rod-like microfossils have been found in cherts and shales from both these rock groups. Analysis of these structures has shown that the outer walls definitely contained carbon compounds and many authorities accept that they are of biological origin; but because of their very simple structure some doubts remain [38], [64], [67]. Undoubted microfossils of diverse and structurally complex microorganisms are relatively abundant in rocks of middle Precambrian age. For example a great wealth of forms is encountered in the Gunflint formation [67], with variations between filamental and spheroidal, between septate and non-septate, all pointing to a varied and intricate morphology, such as is only exhibited by living matter. Other middle and late Precambrian localities where microfossils have been discovered are indicated in Table 3 [38], [67].
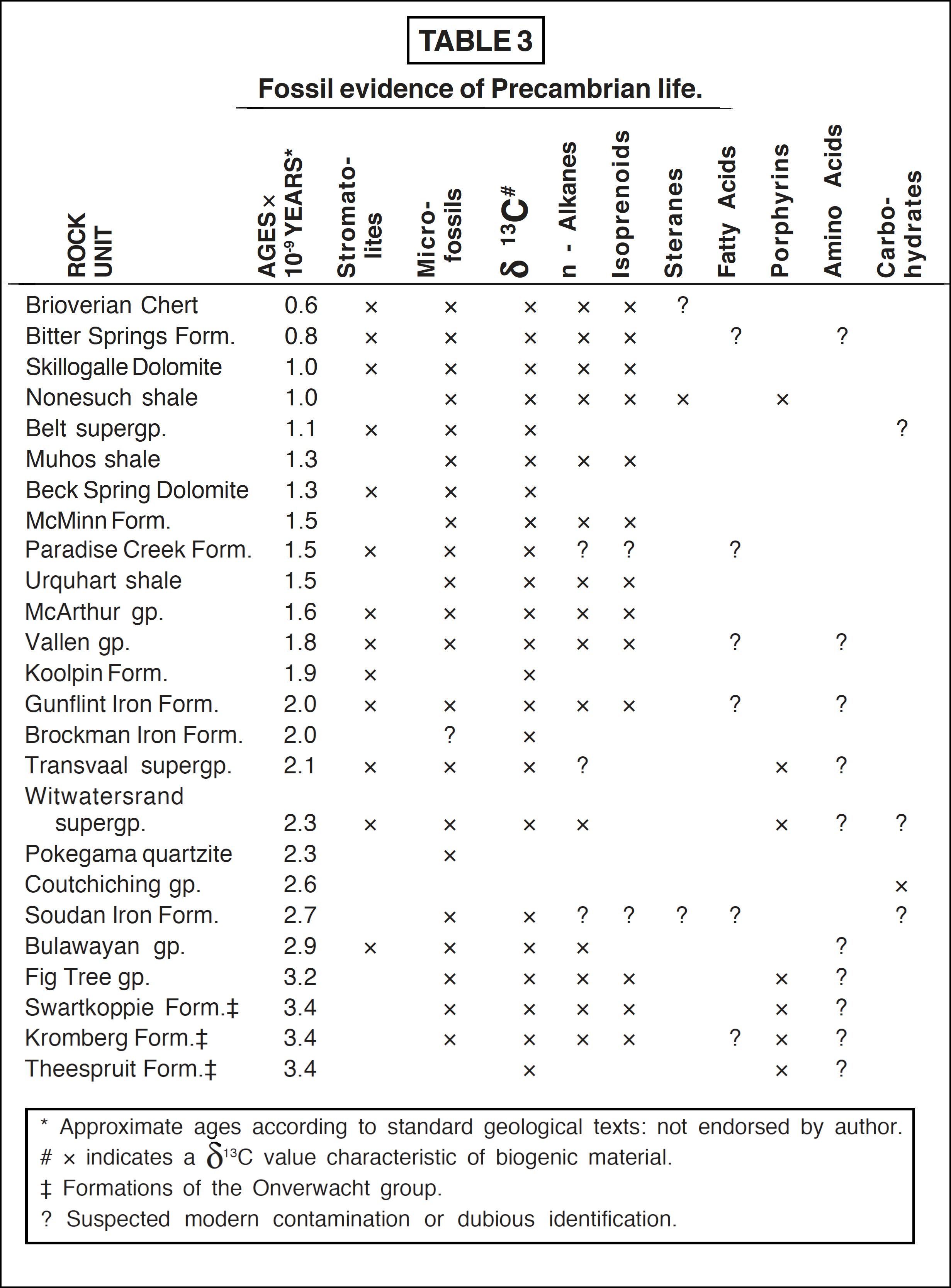
Part of the carbonaceous matter in ancient sediments is extractable by solvents, but most of it is graphitized material (kerogen). A variety of chemical fossils, or "biological markers," have been identified in the extractable fractions from Precambrian cherts and shales. The occurrence of n-alkanes, isoprenoids such as pristane and phytane, steranes, fatty acids, metal chelated porphyrins, amino acids and carbohydrates from the early, middle and late Precambrian formations is indicative of a biological origin for much of the material [68]. The porphyrins, and pristane and phytane, which are possibly degradation products of the side-chains of chlorophylls, are particularly suggestive of photosynthetic organisms. They have been identified in the most ancient sediments from the Onverwacht and Fig Tree series [38]. Amino acids have also been found in rocks throughout the Precambrian [69]; however, serine and threonine, which are known to degrade rapidly on the geological time scale, were also discovered. The amino acids in the Fig Tree and Gunflint formations were also shown to comprise the L-isomers only [70], whereas racemisation is known to be rapid [68]. Contamination from modern sources seems the probable explanation, and the significance of all the chemical fossils is called into question.
The non-extractable kerogen was almost certainly laid down at the time of formation of the host rock. Fractionation of the stable carbon isotopes mass 12 and 13 occurs in living systems and is measured in terms of d13C, negative values indicating material of biological origin and near zero or positive values characterising inorganic carbon-containing remains [71]. In Table 3 are shown the Precambrian formations having negative d13C values characteristic of biogenic material.
Taken together this comprises an impressive array of evidence that life has been present on the earth since the formation of early Precambrian sediments.
C. CONCLUSIONS
The occurrence of a reducing atmosphere on the earth depends on the availability of hydrogen, but hydrogen is known to escape rapidly from the earth's gravitational field because of its low molecular weight. Hydrogen is not a major constituent of the crust at present, and according to the dust cloud theory of the earth's formation, no large reservoir of hydrogen survived consolidation of the earth. Furthermore, there do not appear to be efficient processes for maintaining a flow of hydrogen to the atmosphere, volcanic evolution of hydrogen being minor. The origin and persistence of a reducing atmosphere is therefore difficult to envisage, particularly when the reactivities of reduced gases such as carbon monoxide and ammonia with the ocean are considered.
Oxygen must always have been of major importance in the earth's crust; the composition of the earliest igneous and sedimentary rocks and of the Precambrian ocean, as well as its cosmic and terrestrial abundance, all attest this. Efficient gas-solid reactions are known which transfer oxygen in the form of water, from the vast reservoirs in rocks to the ocean, and two major processes, photolysis of water vapour and photosynthesis, transfer oxygen to the atmosphere. In the light of this evidence, no other conclusion than that oxygen has always been an important constituent of the atmosphere seems possible.
Geological evidence indicates that rocks from the earliest Precambrian are lithologically quite normal and have similar modern counterparts. Weathering, transportation and sedimentation appear to have taken place essentially as at present. Certain sediments containing minerals in reduced form can readily be accounted for in terms of local reducing conditions, such as are found in many areas today, or they are found to be stable in their lower oxidation state sufficiently long enough for erosion, transportation and deposition. There appears to be no persuasive evidence that the atmosphere has ever differed substantially from its present composition. The presence of oxygen in the earth's original atmosphere would, of course, have a dramatic inhibitory effect on the synthesis and accumulation of organic molecules and would virtually rule out the possibility that life arose in this way.
The presence of limestone, and other probably biogenic sediments, of stromatolites, microfossils, chemical fossils and biogenic kerogen in early Precambrian rocks suggests that life originated virtually simultaneously with the formation of the crust of the earth.
ACKNOWLEDGMENTS
The author thanks Professor Lord Tedder, Dr. C. Mitchell and Mr. W.G.C. Walton for reading the typescript and making valuable comments and suggestions.
FOOTNOTES
[1]S.L. Miller and L.E. Orgel. 1974. The origins of life on the earth. Prentice-Hall, Englewood Cliffs, New Jersey, p. 1.
[2]A.I. Oparin. 1924. Proischogdenie Zhizni. Moscovsky Rabotchii, Moscow; 1953. The origin of life. 2nd English edition. Dover Publications, New York.
[3]J.B.S. Haldane. 1929. Science and human life. Rationalist Annual 148:3.
[4]S.L. Miller. 1953. A production of amino acids under possible primitive earth conditions. Science 117:528.
[5]R.M. Lemmon. 1970. Chemical evolution. Chemical Reviews 70:95.
[6]S.W. Fox and K. Dose. 1972. Molecular evolution and the origin of life. W.H. Freeman, San Francisco, chapter 4.
[7]P.H. Abelson. 1956. Amino acids formed in "primitive atmospheres." Science 124:935; 1957. Discussion of S.L. Miller's The formation of organic compounds on the primitive earth. New York Academy of Sciences Annals 69:274; 1966. Chemical events on the primitive earth. Proceedings of the National Academy of Sciences 55:1365.
[8]S.L. Miller and H.C. Urey. 1959. Organic compound synthesis on the primitive earth. Science 130:245.
[9]K. Heyns and K. Pavel. 1957. Zeitschrift fur Naturforschung 12b:97.
[10]R.W. Fairbridge. 1972. Encyclopedia of geochemistry and environmental sciences. Van Nostrand Reinhold, New York.
[11]L.E. Snyder and D. Buhl. 1973. Sky and Telescope 45:156; 1971. Chemical constituents of interstellar clouds. Nature 234:332; D.M. Rank, C.H. Townes and W.J. Welch. 1971. Interstellar molecules and dense clouds. Science 174:1083.
[12]G.A. MacDonald. 1972. Volcanoes. Prentice-Hall, Englewood Cliffs, p. 324; D.E. Whyte and G.A. Waring. 1963. Volcanic emanations. Washington.
[13]See: S.L. Miller and L.E. Orgel, ref. 1, pp. 13-15.
[14]P.H. Abelson. 1966. Chemical events on the primitive earth. Proceedings of the National Academy of Sciences 55:1365.
[15]S.L. Miller and L.E. Orgel, ref. 1, p. 47.
[16]W.A. Fowler, J.L. Greenstein, and F. Hoyle. 1961. Synthesis of deuterons and the light nuclei during the early history of the solar system. American Journal of Physics 29:393.
[17]A. Poldervaart. 1955. Chemistry of the earth's crust. Geological Society of America Special Paper 62:119.
[18]H.C. Urey. 1959. In A.I. Oparin, et al., eds. The Origin of Life on the Earth. Pergamon Press, London, p. 16.
[19]It should be recognised that if this mechanism is efficient, ultraviolet light would also not be available for the synthesis of amino acids etc., except in the upper atmosphere above the oxygen and ozone screen, where they would be particularly vulnerable to decomposition.
[20]L.V. Berkner and L.C. Marshall. 1965. Journal of Atmospheric Science 22:225; 1966. Limitation on oxygen concentration in a primitive planetary atmosphere. Journal of Atmospheric Science 23:133.
[21]R.T. Brinkmann. 1969. Dissociation of water vapour and evolution of oxygen in the terrestrial atmosphere. Journal of Geophysical Research 74:5355.
[22]L. Van Valen. 1971. The history and stability of atmospheric oxygen. Science 171:439.
[23]M.G. Rutten. 1971. The origin of life by natural causes. Elsevier Publishing, Amsterdam.
[24]C.F. Davidson. 1963. The Precambrian atmosphere. Nature 197:893; W.S. Fyfe. 1974. Geochemistry. Clarendon Press, Oxford, pp. 87-94.
[25]K. Rankama. 1955. Geologic evidence of chemical composition of the Precambrian atmosphere. Geological Society of America Special Paper 62:651.
[26]R.H. Nanz. 1953. Chemical composition of pre-Cambrian slates with notes on the geochemical evolution of lutites. Journal of Geology 61:51.
[27]S. Moorbath, R.K. O'Nions, and R.J. Pankhurst. 1973. Early Archean age for the Isua iron formation, West Greenland. Nature 245:138.
[28]C.F. Davidson. 1965. Uranium in ancient conglomerates. Proceedings of the National Academy of Sciences 53:1194; 1964. Geochemical aspects of atmospheric evolution. Economic Geology 59:168.
[29]B.J. Bluck. 1969. Introduction to sedimentology. In G. Eglinton and M.T.J. Murphy, eds. Organic Geochemistry. Springer-Verlag, Berlin, p. 248.
[30]See: S.W. Fox and K. Dose, ref. 6, p. 44.
[31]S.S. Golditch. 1973. Ages of Precambrian banded iron-formations. Economic Geology 68:1126.
[32]P. Ramdohr. 1958. Unweathered pyrite and pitchblende in early Precambrian conglomerates. Abhandlungen der Deutschen Akademie Wissenschaft Berlin, Klasse Chemie, Geologie und Biologie, No. 3.
[33]M.G. Rutten. 1963. The Precambrian atmosphere. Nature 197:893; see also ref. 23.
[34]E.H.P. Cordfunke. 1969. The chemistry of uranium. Elsevier Publishing, Amsterdam.
[35]G. Zeschke. 1960. Transportation of uraninite in the Indus River Pakistan. Transactions of the Geological Society of South Africa 63:87.
[36]See: M.G. Rutten, ref. 23, pp. 260-272.
[37]M. Schidlowski. 1968. Advances in organic geochemistry. Pergamon Press, London, p. 579.
[38]D.M. McKirdy. 1974. Organic geochemistry in Precambrian research. Precambrian Research 1:75.
[39]R.A. Berner. 1970. Sedimentary pyrite formation. American Journal of Science 268:1.
[40]G.H. Taylor. 1971. Society of Mineralogy and Geology of Japan. Special Issue No. 3, p. 283.
[41]G.M. Koen. 1961. The genetic significance of the size distribution of uraninite in Witwatersrand blankets. Transactions of the Geological Society of South Africa 64:23.
[42]R.P. Viljoen, R. Saager, and M.J. Viljoen. 1970. Mineralium Deposits 5:164.
[43]G.H. Taylor, quoted in ref. 38, p. 119.
[44]P. Cloud. 1974. Evolution of ecosystems. American Scientist 62:54.
[45]M.G. Rutten, ref. 23, p. 276.
[46]See: H. Lepp and S.S. Golditch. 1964. The origin of Precambrian iron formations. Economic Geology 59:1025.
[47]J.L. Hough. 1958. Fresh-water environment of deposition of Precambrian banded iron formations. Journal of Sedimentary Petrology 28:414.
[48]G.J.S. Govett. 1966. Origin of banded iron formations. Geological Society of America Bulletin 77:1191.
[49]F.B. Van Houten. 1964. Origin of red beds some unsolved problems. In A.E.M. Nairn. Problems in Palaeoclimatology. John Wiley, New York, p. 647.
[50]W.S. Fyfe, ref. 24, p. 99.
[51]P.E. Cloud. 1969. Pre-Paleozoic sediments and their significance for organic geochemistry. In G. Eglinton and M.T.J. Murphy, eds. Organic Geochemistry. Springer-Verlag, Berlin, p. 727.
[52]F.J. Pettijohn. 1943. Archean sedimentation. Geological Society of America Bulletin 54:925.
[53]P.E. Cloud. 1968. Atmospheric and hydrospheric evolution on the primitive earth. Science 160:729.
[54]G.E. Murray. 1965. American Association of Petroleum Geologists Bulletin 49:3; T.R. Wildman and L.A. Haskin. 1973. Rare earths in Precambrian sediments. Geochimica et Cosmochimica Acta 37:419, 439.
[55]A.V. Sidorenko. 1969. Doklady Akademiya Nauk SSSR 186:36.
[56]L.G. Sillen. 1967. How have sea water and air got their present compositions? Chemistry in Britain 3:291.
[57]M.G. Rutten, ref. 23, pp. 311, 319.
[58]M.G. Rutten, ref. 23, p. 254.
[59]A.M. MacGregor. 1940. Transactions of the Geological Society of South Africa 43:9.
[60]R.B. Young. 1940. Transactions of the Geological Society of South Africa 43:17.
[61]M. Härme and V. Perttunen. 1963. Comptes Rendus des Sèances de la Société Geologique de Finlande 35:79.
[62]B. Nagy. 1970. Porosity and permeability of early Precambrian Onverwacht chert: origin of the hydrocarbon content. Geochimica et Cosmochimica Acta 34:525.
[63]S. Sanyal, S.S. Marsden, and K.A. Kvenvolden. 1971. Permeabilities of Precambrian Onverwacht cherts. Nature 232:325.
[64]J.W. Schopf. 1975. Precambrian paleobiology: problems and perspectives. Annual Review of Earth and Planetary Science 3:213; 1975. The age of microscopic life. Endeavour (I.C.I. magazine) 34:51.
[65]See also: M.G. Rutten, ref. 23, p. 246, for other possible macrofossils.
[66]E.S. Barghoorn and S.A. Tyler. 1965. Microorganisms from the Gunflint chert. Science 147:563.
[67]J.W. Schopf. 1970. Biological Reviews 45:323.
[68]G. Eglinton and M.T.J. Murphy, eds. 1969. Organic geochemistry. Springer-Verlag, Berlin.
[69]K.A. Kvenvolden. 1975. Geochemistry of amino acids. Annual Review of Earth and Planetary Science 3:18.
[70]P.H. Abelson and P.E. Hare. 1969. Carnegie Institute of Washington Yearbook 67:208; K.A. Kvenvolden, E. Peterson, and G.E. Pollock. 1969. Optical configuration of amino acids in Precambrian Fig Tree chert. Nature 221:141; J. Oró, et al. 1971. Configuration of amino-acids in carbonaceous chondrites and a Precambrian chert. Nature 230:107.
[71]E.T. Degens. 1969. Biogeochemistry of stable carbon isotopes. In G. Eglinton and M.T.J. Murphy, eds. Organic Geochemistry. Springer-Verlag, Berlin, p. 304.
[72]F.J. Pettijohn. 1975. Sedimentary rocks. 3rd edition. Harper and Row, New York, p. 588.
[73]G.R. Carruthers and T. Page. 1972. Apollo 16 far-ultraviolet camera-spectrograph: earth observations. Science 177:788; see also G.E. Snow and G.T. Javor. 1975. Oxygen and evolution. ORIGINS 2:59, for further comments.